 Thus, it takes much more energy than just overcoming a larger ionic charge would suggest. This is the energy per mole necessary to remove electrons from gaseous atoms or atomic ions. These tables list values of molar ionization energies, measured in kJmol 1. As you go from left to right, you go from low ionization energy to high ionization energy. Molar ionization energies of the elements. Ca has a higher IE 1 because its radius is smaller. The third ionization energy for the ion formed was found to be 2,700 kJ/mol. The second ionization energy for the ion formed was found to be 1,800 kJ/mol. 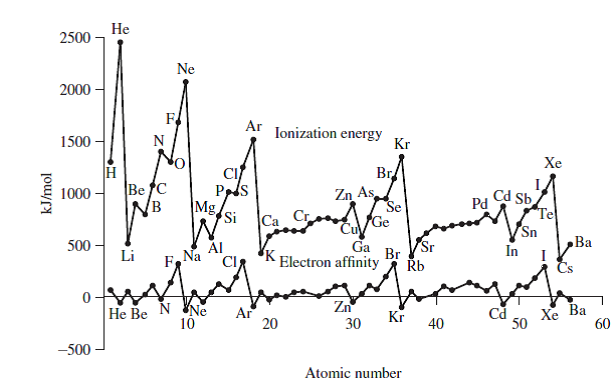
So, this is high, high ionization energy, and thats the general trend across the periodic table. Using experimental data, the first ionization energy for an element was found to be 600 kJ/mol. Of C or P, the element with the higher first ionization energy is Of Ca or Sr, the element with the higher first ionization energy is Of Sn or I, the element with the higher first ionization energy is Of F or S, the element with the higher first ionization energy is. Both of these are properties of the isolated atom. Make certain each sentence is complete before submitting your answer. SAMPLE PROBLEM 8.4 Ranking Elements by First Ionization Energy. Why is it so much larger? Because the first two electrons are removed from the 3 s subshell, but the third electron has to be removed from the n = 2 shell (specifically, the 2 p subshell, which is lower in energy than the n = 3 shell). Or especially the first electron, and then here you have a high ionization energy. Electronegativity is a function of: (1) the atom's ionization energy (how strongly the atom holds on to its own electrons) and (2) the atom's electron affinity (how strongly the atom attracts other electrons). Plan To rank the elements by atomic size, we find them in the.  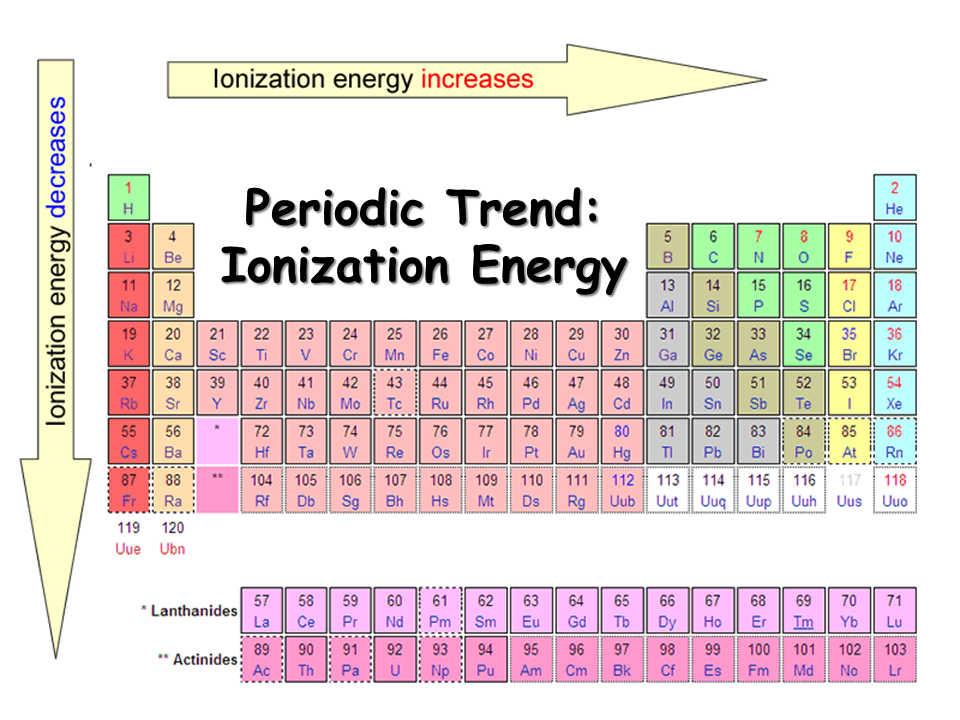
The third IE, however, is over five times the previous one. 3) Mg Ca Sr Ba a) Most metallic b) Least metallic. The second IE is twice the first, which is not a surprise: the first IE involves removing an electron from a neutral atom, while the second one involves removing an electron from a positive ion. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
